Monoamine Oxidase project
Master project in computational and medicinal chemistry (2013 - 2015)
University of Chile, Faculty of Chemical and Pharmaceutical Science

Monoamine oxidase inhibitors (MAOIs) are interesting compounds due to their potential applicability in psychiatric and neurodegenerative diseases such as depression, Alzheimer and Parkinson diseases. Even though Phenylalkylamines have structural and chemical commonalities they may behave differently upon binding in the active site of MAO A and B isoforms. According to experimental results of para-chloro-beta-methylphenylethylamine (BME) and para-methoxy-beta-methylphenylethylamine (PMB) on MAO-B display distinct inhibitory and substrate activities, respectively.
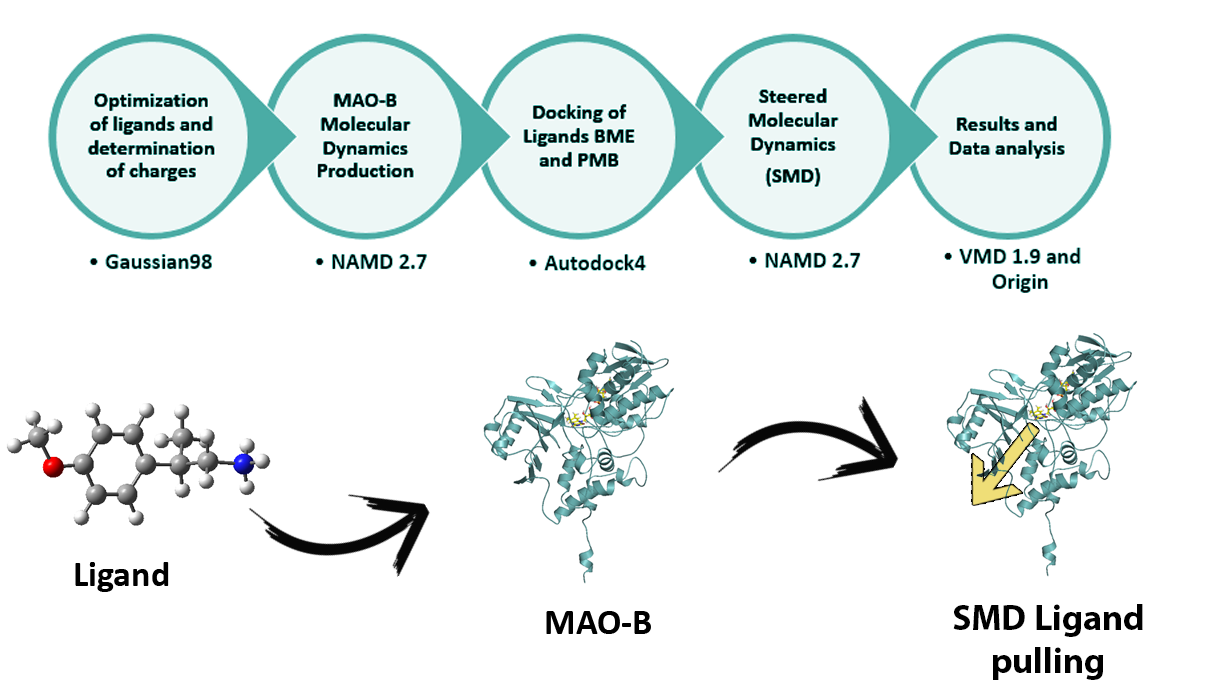
In this project the interactions of these two phenylalkylamine derivatives were studied using MAO-B combining different approaches and methodologies such as Quantum calculations, ligand-protein docking and advance Molecular Dynamics simulations techniques. By means of docking studies I determined the orientations and probable interactions at the described binding site. Then, the entrance cavities were explored using Steered Molecular Dynamics (SMD) pulling the ligands from the binding cavity towards the exit of the enzyme aiming to shed light on their odd displayed activity.
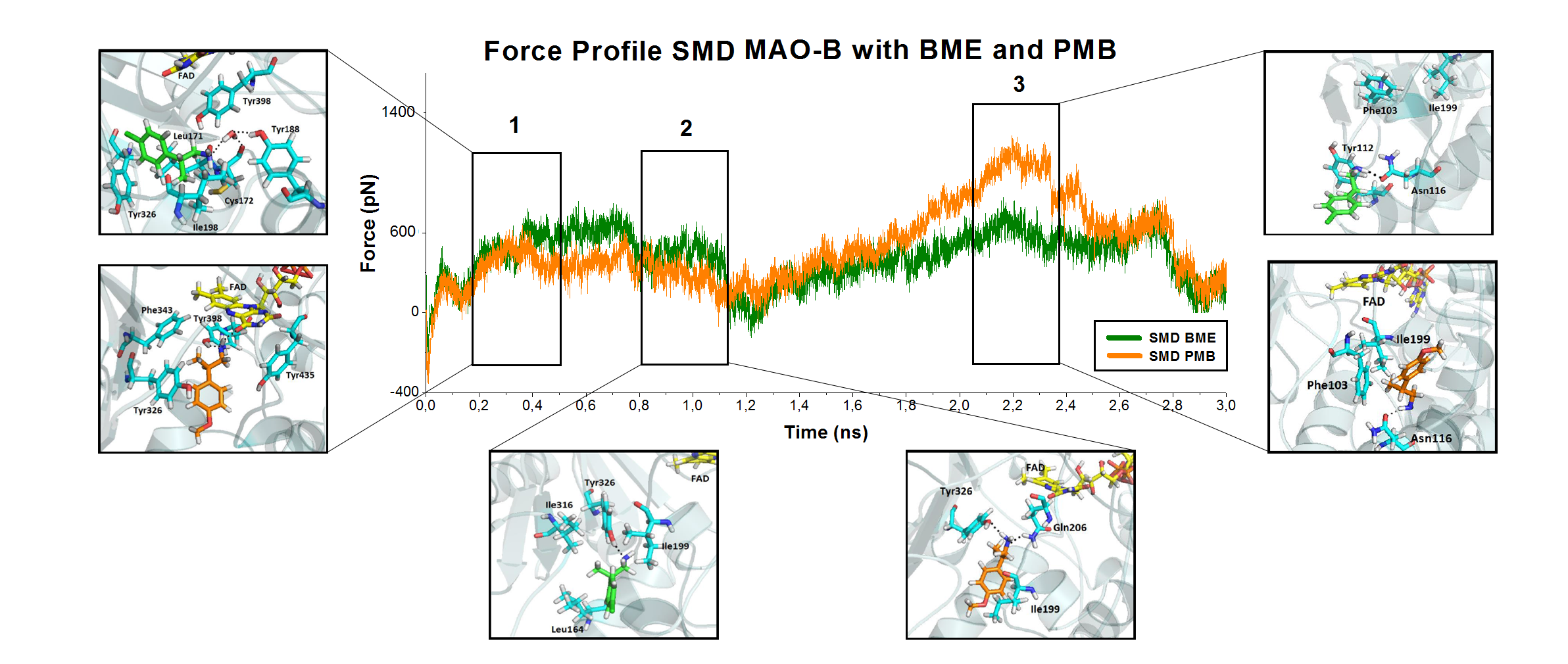
Using SMD the ligands were allowed to move from the binding site to the exit of the protein at constant speed. This methodology allow us to study the behaviour of these compounds going from the entrance cavity towards the binding site aiming to identify key interactions that might be responsible for their displayed activities on MAO-B. The study of the interactions and their associated energies allowed us to identify three zones on the force profiles. Comparing these three areas, I noticed important differences in both compounds identifying certain residues at the entrance cavity that would be responsible in the recognition of MAO-B ligands.